




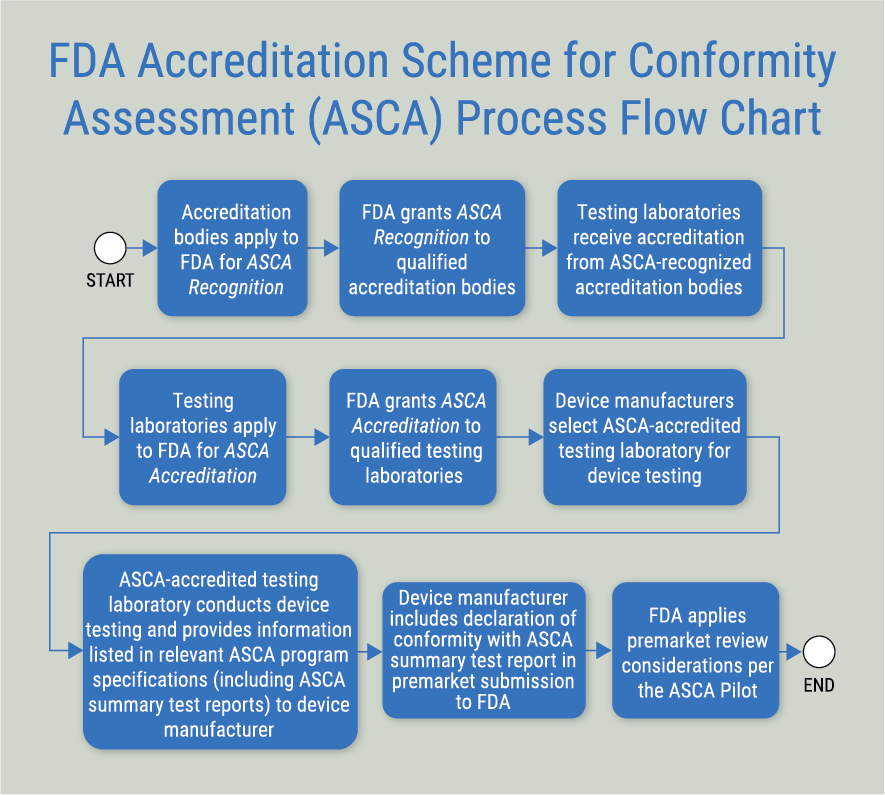
The first step in the process is to obtain accreditation from an ASCA-recognized accreditation body (i.e. IAS), which includes an assessment to ISO/IEC 17025 and the ASCA program specifications. For laboratories already accredited by IAS to ISO/IEC 17025, the process involves a scope expansion by IAS to include the ASCA program specifications. The next step involves the laboratory submittal of an application for ASCA Accreditation to the FDA.
Join the FDA/ASCA List
Get the latest information about accreditation practices, standards, training courses and more, when you join the IAS list for FDA/ASCA. To join, click here.
As of September 19, 2023, ASCA has become a Permanent Program – Read More
ASCA Pilot Guidances
Website Resources
ASCA Pilot Web page
Standards & Conformity Assessment Program
FDA Recognized Consensus Standards Database
www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfStandards/search.cfm
Standards Resources
Recognition and Withdrawal of Voluntary Consensus Standards, final guidance
Appropriate Use of Voluntary Consensus Standards in Premarket Submissions for Medical Devices, final guidance
CDRH Learn: How to Study and Market Your Device: Standards
www.fda.gov/training/cdrhlearn/default.htm
Industry Education:
Three Resources for You
CDRH Learn: Multi-Media Industry Education
Over 200 modules
Videos, audio recordings, power point presentations, software-based “how to” modules
Mobile-friendly: access CDRH Learn on your portable devices
Device Advice: Text-Based Education
Comprehensive regulatory information on premarket and postmarket topics
Division of Industry and Consumer Education (DICE)
Contact DICE if you have a question
Email: DICE@fda.hhs.gov
Phone: 1(800) 638-2041 or (301) 796-7100 (Hours: 9 am-12:30 pm; 1 pm-4:30pm EST)
Web: www.fda.gov/DICE
Basic Safety and Essential Performance
| ANSI/AAMI ES60601-1 | IEC 60601-2-11 | IEC 60601-2-36 | ISO 80601-2-12 |
| IEC 61010-1 | IEC 60601-2-16 | IEC 60601-2-37 | ISO 80601-2-13 |
| IEC 60601-1-2 | IEC 60601-2-17 | IEC 60601-2-43 | IEC 80601-2-30 |
| IEC 60601-1-3 | IEC 60601-2-18 | IEC 60601-2-44 | IEC 80601-2-35 |
| IEC 60601-1-6 | IEC 60601-2-19 | IEC 60601-2-45 | ISO 80601-2-55 |
| IEC 60601-1-8 | IEC 60601-2-20 | IEC 60601-2-47 | ISO 80601-2-56 |
| IEC 60601-1-10 | IEC 60601-2-21 | IEC 60601-2-50 | IEC 80601-2-59 |
| IEC 60601-1-11 | IEC 60601-2-22 | IEC 60601-2-52 | IEC 80601-2-60 |
| ANSI/AAMI HA60601-1-11 | IEC 60601-2-23 | IEC 60601-2-54 | ISO 80601-2-61 |
| IEC 60601-1-12 | IEC 60601-2-25 | IEC 60601-2-57 | ISO 80601-2-69 |
| IEC 60601-2-1 | IEC 60601-2-27 | IEC 60601-2-62 | ISO 80601-2-70 |
| IEC 60601-2-2 | IEC 60601-2-28 | IEC 60601-2-63 | ISO 80601-2-72 |
| IEC 60601-2-5 | IEC 60601-2-29 | IEC 60601-2-64 | ISO 80601-2-74 |
| IEC 60601-2-6 | IEC 60601-2-31 | IEC 60601-2-65 | IEC 80601-2-77 |
| IEC 60601-2-8 | IEC 60601-2-33 | IEC 60601-2-68 | ISO 80601-2-79 |
| IEC 60601-2-10 | IEC 60601-2-34 | IEC/TR IEC 60601-4-2 | ISO 80601-2-80 |
To convey your laboratories’ interest in the program to IAS, please email: